Stomach Cancer Causes, Symptoms, Surgery, Chemotherapy, Prognosis
What is Stomach Cancer?
Stomach cancer or gastric cancer is a malignant tumor that develops in the stomach. Although there may be benign growths like certain types of stomach polyps, most of the tumors that develop in the stomach are malignant tumors. Generally the term gastric cancer refers to an adenocarcinoma of stomach as it is the most common form of malignant tumor of the stomach.
Signs and Symptoms
Early gastric cancer is often asymptomatic or associated with vague symptoms which can delay the diagnosis in many patients. It is often mistaken for other gastrointestinal conditions in these early stages and without proper diagnostic investigation, it can remain undiagnosed until the generalized symptoms associated with cancer prompts further investigation.
Weight loss is one of the most common symptoms associated with later stages of gastric cancer. Other general symptoms include :
- loss of appetite
- weakness
- epigastric pain, not relieved by antacids, and pain may radiate to the back with pancreas involvement
- difficulty swallowing food (dysphagia)
- generalized abdominal discomfort
A tumor of gastroesophageal junction or in the fundus of stomach is often associated with dysphagia. Tumor of the distal-end may cause partial obstruction to the outflow of gastric contents and can present with persistent vomiting. Vomiting can also be seen in patients with diffuse gastric tumors affecting the effective gastric contractions. A diffuse tumor can also prevent distension the stomach wall, resulting in feeling of fullness of stomach with minimal food (early satiety).
Occasionally some patients can have significant gastrointestinal bleeding which is often associated with vomiting of blood (hematemesis) or black tarry stools (melena) due to passing of the blood into the lower gastrointestinal tract.
Symptoms of Stomach Cancer Spread
Signs of metastasis suggest incurable or an inoperable stage. Gastric cancers that metastasize to liver can cause jaundice or pain over the liver. Sometimes the tumor mass can be felt on examination. Cough, hiccoughs, or coughing up blood (hemoptysis) is seen in patients with gastric cancer that has spread to lungs.
Spread of cancer into the peritoneal cavity can lead to malignant ascites or a malignant mass in the ovary (called Krukenberg’s tumor). Sometimes cancer can spread to the subcutaneous tissue around umbilicus to form a nodular swelling near the umbilicus (called Sister Mary Joseph’s node). Lymphatic spread can result in enlargement of nodes above the clavicle on the left side (called Virchow’s node).
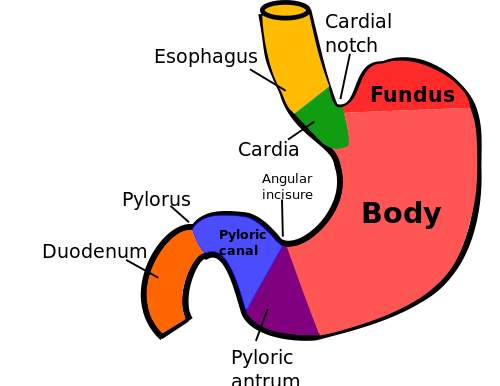
Causes of Stomach Cancer
The exact cause of stomach cancer is unknown but this type of malignancy is more likely to arise in a person with one or more of the risk factors.
One of the more widely accepted explanations of stomach cancer revolves around chronic H.pylori infection. Persistent inflammation, which is often seen with chronic H.pylori infection leads to atrophic gastritis. This reduces the acidity of the stomach juices (achlorhydria) and allows for other bacterial strains to thrive in this environment. Some of these bacteria are capable of converting dietary nitrates to nitrites and can lead to the formation of toxic N-nitroso compounds.
These compounds in combination with other risk factors, like genetic factors, cigarette smoking and so on, produce genetic mutations and abnormal proliferation of the stomach cells. This can lead to metaplasia and dysplasia of the gastric epithelial cells lining the stomach which eventually results in cancer. Other risk factors, even in the absence of H.pylori infection, may also lead to genetic mutations of the stomach cells which can ultimately contribute to cancer development.
Risk Factors
Stomach cancer affects men more than women and is more often seen in the over 50 age group. Overall, stomach cancer is considered to be an outcome of multiple factors. Several risk factors have been identified from epidemiological studies and can be grouped as acquired factors, genetic factors and precancerous conditions.
Acquired
- Diet/nutritional factors
- high salt consumption (salted or pickled foods)
- excessive smoked foods (like smoked fish)
- high nitrates/nitrites consumption
- high carbohydrates intake
- lack of fresh fruit and vegetables in diet
- low dietary vitamin A and C
- Infections
- Helicobacter pylori (H.pylori)
- Epstein-Barr virus (EBV)
- Cigarette smoking
- Poor quality drinking water
- Occupational risk factors
- Coal
- Rubber
- Exposure to radiation
- Prior gastric surgery for benign gastric ulcer disease
Genetic
- Family history of stomach cancer
- Pernicious anemia
- Hereditary nonpolyposis colorectal cancer
Precancerous
- Chronic atrophic gastritis
- Adenomatous gastric polyps of 2 cm or more
- Pernicious anemia
- Intestinal metaplasia
- Gastric dysplasia
- Hypertrophic gastropathy (Menetrier’s disease)
- Chronic gastric ulcer (stomach ulcers)
- Stump of previous gastric removal surgery
Other risk factors are more significant for cancer at the gastroesophageal junction or cardiac end of stomach and includes :
- high calorie intake
- increased body mass index (obesity)
- chronic gastroesophageal reflux disease (GERD)
Types of Stomach Cancer
An adenocarcinoma is the most common type of stomach cancer accounting for over 90% of all malignant stomach tumors. Other rare malignant cancers include leiomyosarcoma, squamous cell carcinoma, and carcinoid tumors. Lymphomas are also seen in stomach.
The gastric cancers can be broadly grouped based on the appearance of the cancer lesion (Borrmann classification). This includes :
- Type I – polypoid or fungating type
- Type II – ulcer type with elevated borders
- Type III – infiltrating ulcer type
- Type IV – diffuse infiltrating type
- Type V – unclassified group
Based on the histology, well differentiated cancers may be grouped as the intestinal type which shows a tendency to form glands and the poorly differentiated cancers as diffuse type. The diffuse type shows widespread thickening of the stomach and can sometimes form a non-distensible stomach known as linitis plastica. The diffuse type is more aggressive and spreads faster than the intestinal type.
Younger patients are often affected by the diffuse type and has poor prognosis compared to the intestinal type. The intestinal type is more frequent in older men and it arises from the precancerous conditions like atrophic gastritis. Intestinal type is common in the regions with high incidence of gastric cancer while diffuse type is more common in low-incidence regions. The diffuse type is seen in familial forms of gastric cancer and also associated with the blood group A.
The gastroesophageal junction cancers are of 3 types (Siewert classification). These are :
- Type I
- The tumor arises from the distal end of esophagus and progresses to the gastroesophageal junction below.
- The tumor usually develops following intestinal metaplasia of the lower end of esophagus.
- Barrett’s esophagus is a precancerous condition resulting from chronic GERD characterized by intestinal metaplasia at the lower end of the esophagus.
- Type II
- The tumor arises from the cardiac end or from gastroesophageal junction.
- Type III
- Tumor arises from below the cardiac end of stomach and spread from below to the gastroesophageal junction.
Stomach Cancer Diagnosis
Physical examination
Stomach cancer (gastric cancer) is asymptomatic (no symptoms /silent) in the early stages and physical signs may be absent. As the cancer advances, a patient reports unintentional weight loss and cachexia. Depending on the size of the tumor, a mass may be palpated in the epigastric region (upper middle abdominal areas).
One of or more of the stomach cancer symptoms may also be present and reported by patients. The severity of these symptoms can vary and it is not uncommon for other gastrointestinal conditions to be initially diagnosed. The presentation may also differ slightly depending on the type of stomach cancer.
Laboratory investigations
- Iron deficiency anemia may be found on routine hematological studies in the gastric cancer patients.
- Pernicious anemia may progress to megaloblastic anemia and this may also be found on hematological investigation.
- The malnourishment seen in gastric cancer patients may be seen as low blood levels of albumin (hypoalbuminemia).
- Stools test for occult blood may be positive.
Endoscopy
Endoscopy is the best method to diagnose gastric cancer as it allows direct visualization of the gastric mucosa and a biopsy of tissue can be sent for histopathologic evaluation. Endoscopy with biopsy and histopathologic examination has a diagnostic accuracy of 95 to 99%.
Ultrasound
Endoscopic ultrasonography (EUS) may be used to stage gastric cancer after confirming the diagnosis. It may also be helpful in diagnosing diffuse-type gastric carcinoma in some patients. The EUS helps in assessment of the extent of tumor, invasion of the stomach wall and lymph node involvement. EUS findings can complement the findings of other staging studies like the CT scan.
Tumor Markers
Tumor markers like carcinoembryonic antigen (CEA), CA 72.4, CA 19-9 or CA 50 can also be of use in correlating with the stage of the cancer. These tests are also of value post-operatively to monitor for any recurrence.
CT Scan
Computerized tomography (CT) scan of the abdomen and pelvis is an important non-invasive test for the staging of gastric cancer. CT scan is useful in assessing the lymph node involvement, peritoneal spread of the tumor, metastasis to liver or pancreas and the degree of tumor involvement of the gastric wall.
A CT scan of the chest should also be performed to record the lymph node status in the chest and to rule out spread to the lungs. Positron emission tomography (PET) scan may be of use in staging of disease in some patients especially those with metastasis beyond peritoneal cavity.
Laparoscopy
Staging laparoscopy is sometimes done before the planning the treatment in patients who have been found to have localized gastric cancer on CT scan. The laparoscopy aims to identify any small low-volume metastases into peritoneal cavity that may not be detected on CT. Additionally a peritoneal cytology can also be done during laparoscopic procedure to detect presence of cancer cells in the peritoneal cavity.
Screening for Stomach Cancer
Radiological studies, like a double-contrast barium radiograph or upper gastrointestinal endoscopy, are effective for stomach cancer screening. The lesion in gastric cancer, namely the ulcer or the growth can be first noted in the upper gastrointestinal barium double-contrast study. An upper GI endoscopy and biopsy should then be conducted. Screening should be considered for individuals who are considered as high risk.
Staging of Stomach Cancer
Staging of gastric cancer is essential for assessing the outcome and to predict the prognosis with reasonable accuracy. To have a uniform staging system the American Joint Committee on Cancer (AJCC) system is commonly used. Alternatively the Japanese system may be used for staging. Read more on Cancer Staging.
The tumor (T) stage in the AJCC staging system is determined by assessing the depth of invasion of the tumor into the gastric wall and the invasion into adjacent structures.
- T1 – tumor invading lamina propria or submucosa layers of the stomach
- T2 – tumor invading muscularis propria is T2a or subserosa is T2b
- T3 – tumor penetrating visceral peritoneum (or serosa) without invasion of adjacent structures
- T4 – tumor invading adjacent structures
The Nodal staging (N) is done by assessing the number of regional lymph nodes involved, irrespective of the location.
- N1 – metastasis to 1 to 6 regional lymph nodes
- N2 – metastasis to 7 to 15 regional lymph nodes
- N3 – metastases to more than 15 regional lymph nodes
Metastasis staging is similar to other cancers with M0 for the absence of distant metastasis and M1 for presence of distant metastasis.
The amount of residual disease that remains after the tumor resection (surgical removal) is staged using the R classification system.
- R0 – no gross or microscopic residual disease
- R1 – microscopic residual disease
- R2 – gross residual disease
Surgery
Stage I Gastric Cancer (Early Stomach Cancer)
Surgical removal (resection) of the tumor is the only chance for a possible cure for stomach cancer. It is the best option in early stage gastric cancer. Traditionally surgery may involve limited resection or gastrectomy. Endoscopic mucosal resection (EMR) is a new procedure that can spare the resection of stomach.
Endoscopic Mucosal Resection (EMR)
Patients with well-differentiated, small superficial lesions with minimal potential for spread can undergo an EMR with complete resection of the tumor without resection of the stomach or lymph nodes. It involves injection of fluid into the submucosa of the stomach to elevate the affected area. A complete mucosal resection of the lesion is then performed endoscopically. EMR is not suitable for a tumor that has invaded the submucosa as there is a high risk of spread to lymph nodes.
Limited Resection of the Stomach
Limited resection of the stomach is an alternative option if EMR is not viable, thereby avoiding gastrectomy. This procedure is considered for small lesions where the full thickness of the stomach wall with along with the complete lesion is removed.
Gastrectomy
Gastrectomy with lymph node dissection is the main option if limited resection or EMR is not viable. A tumor limited to the lower (pyloric or distal) end of the stomach can be treated with a subtotal gastrectomy and lymph node surgery. Tumors of the proximal end (cardiac end) of the stomach require total gastrectomy with lymph node removal. For some cancers of the gastroesophageal junction, esophagectomy (removal of the esophagus) may also be necessary while some can be managed with removal of the stomach and the abdominal part of esophagus (extended gastrectomy).
Stage II and III Gastric Cancer
Gastrectomy with lymph node removal surgery is the mainstay of treatment in all patients. In addition partial removal of the pancreas or spleen may be required in some patients. Surgical resection alone may not be sufficient for curing the cancer in most patients.
Pre-operative neoadjuvant chemotherapy (administered before surgery) and post-operative adjuvant chemotherapy (administered after surgery) is often employed in addition to surgery to reduce the high risk of recurrence following surgery alone. The chemotherapy agents commonly used for adjuvant or neoadjuvant therapy are various combinations containing agents like :
- epirubicin
- cisplatin
- mitomycin C
- fluorouracil
- floxuridine
Adjuvant therapy with systemic chemotherapy following surgery has shown some benefit in preventing or delaying recurrence and improves the survival rate. Sometimes the adjuvant chemotherapy may be given intraperitoneally immediately after surgical resection of stomach in patients with high risk of intraperitoneal spread. Recently a combination of chemotherapy (fluorouracil and leucovorin containing regimen) with radiotherapy (chemoradiotherapy) is being used post-operatively with greater success.
Neoadjuvant chemotherapy can be of help in decreasing the stage of the primary cancer and thereby increase the possibility of complete resection of the primary tumor. It also provides an option for early treatment of microscopic metastatic lesions.
Advanced Gastric Cancer
Chemotherapy with best supportive care should be considered for patients with advanced gastric cancer, if they can tolerate it. It offers a better survival advantage than supportive care alone.
Chemotherapy
Single-drug chemotherapy may be adopted in patients with poor performance status. Drugs known to produce partial response like 5-fluorouracil, mitomycin C, or cisplatin may be used for single-drug therapy. Other possible drugs that may be of use in single-drug therapy includes capecitabine, docetaxel, irinotecan, and epirubicin. The single-drug treatments produce response rates of 10% to 25% however the response is often short-lived.
Combination therapy has been found to have modest improvement in survival, compared to the single-drug therapy and hence combination therapy may be administered in patients who can tolerate it. Commonly employed combination therapy regimens include cisplatin-fluorouracil (CF), epirubicin, cisplatin and flurouracil (ECF) and docetaxel, cisplatin and flurouracil (DCF). Other newer regimens considered for gastric cancer include irinotecan-fluorouracil-leucovorin (FOLFIRI) and fluorouracil-leucovorin-oxaliplatin (FOLFOX).
Palliative Care
Palliative Surgery
In a patient with incurable gastric cancer with a poor prognosis, palliative surgeries are done to provide symptomatic relief and improve the quality of life. The frequent symptoms for which patients undergo surgery includes pain, bleeding, gastric or esophageal obstruction and vomiting. The surgeries performed include :
- gastrojejunostomy – connecting the stomach and jejunum to overcome obstruction of stomach
- palliative gastrectomy – removal of the stomach
- gastrostomy – opening of stomach externally to abdominal wall for feeding
Palliative Radiation Therapy
The role of radiotherapy in advanced gastric cancer is limited to palliative radiotherapy. It may be used for easing of symptoms like bleeding, obstruction or pain.
Complications of Treatment
The treatment of stomach cancer is associated with several immediate and long term complications. Surgical resection is associated with bleeding, leakage from the anastomosis (joining of gut following gastrectomy), and damage to surrounding structures. Palliative surgeries are can also end up with fatal anastomotic leak.
A long term complication of surgical removal of the stomach is dumping syndrome, which is characterized by diarrhea, abdominal cramps and sometimes palpitations following a meal. This usually results from a sudden release of some gastrointestinal hormones due to rapid movement of the ingested food to the intestines. Long term complications can also result in malabsorption of vitamin B12, iron and calcium, which may require monitoring and supplementation.
Chemotherapy is associated with a wide range of adverse effect including :
- mucositis
- diarrhea
- hair loss
- edema (swelling)
- bone marrow suppression (neutropenia, low platelet counts)
- febrile neutropenia
Drugs like docetaxel are known to cause allergic reactions. Cisplatin is associated with severe nausea and vomiting and oxaliplatin is associated with peripheral neuropathy. Toxicity of combination chemotherapy is slightly higher than the single-drug therapy and is also associated with higher treatment-related mortality. Some regimens like irinotecan-based and oxaliplatin-based are less toxic compared to the cisplatin or flurouracil-based regimens.
Stomach Cancer Prognosis
The prognosis for stomach cancer depends on the stage at which the diagnosis is made. An earlier diagnosis translates into a better prognosis. Good screening systems are the cornerstone of early diagnosis and high risk patients should consider undergoing regular screening. Surgical resection of the tumor at an early stage is the only curative treatment for gastric cancer. Additional treatment given in the form of adjuvant or neoadjuvant chemotherapy can further improve the results.
Approximately 30% of patients undergoing curative surgical resection survive beyond 5 years. The overall 5-year survival rate including all patients across all stages of gastric cancer is less than 10%. Surgical resection may be curative in early gastric cancer confined to the mucosa and submucosa. Anatomic location is another important prognostic factor. The cancers of the distal end of the stomach have a better prognosis than cancers of the proximal or the cardiac end of stomach. Prognosis is relatively poor for the diffuse-type in general and especially in those with linitis plastica. Other factors affecting the prognosis of stomach cancer include age, tumor size and depth.
In countries like Japan where mass screening techniques are employed due to the high incidence of stomach cancer, tumors are detected while still confined to the mucosa and submucosa. The 5 year survival rate increases sharply, as high as 50%, due to early detection. However, the most important factor affecting the prognosis is the stage of the cancer and lymph node status (number of positive lymph nodes).
References
- Gastric cancer. Medscape
- Stomach cancer. Cancer.gov
Last updated on September 1, 2018.